   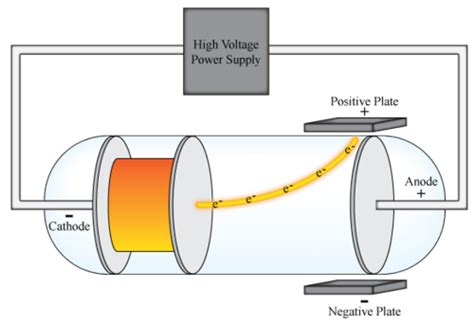
W ilhelm Conrad Röntgen was born on March 27, 1845, at Lennep in the Lower Rhine Province of Germany, as the only child of a merchant in, and manufacturer of, cloth. Share via Email: Wilhelm Conrad Röntgen – Biographical Share this content via Email.Share on LinkedIn: Wilhelm Conrad Röntgen – Biographical Share this content on LinkedIn.Tweet: Wilhelm Conrad Röntgen – Biographical Share this content on Twitter.Share on Facebook: Wilhelm Conrad Röntgen – Biographical Share this content on Facebook.Quarks are elementary because quarks cannot be broken down any further. Protons and neutrons are also not elementary particles because they are made up of even smaller particles called quarks joined together by other particles called gluons (because they "glue" the quarks together in the atom). Some idea of present-day atomic physics can be found in the links in the table below.Ītoms are not elementary particles, because they are made of subatomic particles like protons and neutrons. Although this model was well understood, modern physics has developed further, and present-day ideas cannot be made easy to understand. Isotopes vary in the number of neutrons present in the nucleus. This experiment was called the Geiger–Marsden experiment or the Gold Foil Experimentīy this stage the main elements of the atom were clear, plus the discovery that atoms of an element may occur in isotopes. Rutherford showed this when he used an alpha radiation source (from helium) to hit the very thin gold sheets, surrounded by a Zinc sulphide lampshade that produced visible light when hit by alpha emissions. In 1910, the New Zealand physicist Ernest Rutherford put forward the idea that the positive charges of the atom were found mostly in its center, in the nucleus, and the electrons ( e-) around it. Lord Ernest Rutherford later named these positively charged particles protons Rutherford's atomic modelĪtomic experiment of Lord Ernest Rutherford In the same time, experiments by Eugene Goldstein in 1886 with cathode discharge tubes allowed him to establish that the positive charges had a mass of 1.6726 * 10 −27 kg and an electrical charge of +1,6 * 10 −19 C. In 1906, Robert Millikan determined that the electrons had a Coulomb (C) charge of -1.6 * 10 −19, something that allowed calculation of its mass as tiny, equal to 9.109 * 10 −31 kg. The negative charges were named electrons ( e-).Īccording to the assumptions established about the atoms neutral charge, Thomson proposed the first atomic model, that was described as a positively-charged sphere in which the electrons were inlaid (with negative charges). Thomson knew that the atoms were electrically neutral, but he established that, for this to occur, an atom should have the same quantity of negative and positive charges. Crookes named the emission ' cathode rays'.Īfter the cathode ray experiments, Sir Joseph John Thomson established that the emitted ray was formed by negative charges, because they were attracted by the positive pole. When creating a vacuum in the tube, a light discharge can be seen that goes from the cathode (negatively-charged electrode) to the anode (positively-charged electrode). In 1850, Sir William Crookes constructed a ' discharge tube', that is a glass tube with the air removed and metallic electrodes at its ends, connected to a high voltage source. Schematic representation of the Thomson model. Chemical changes correspond to a reorganisation of the atoms taking part in the chemical reaction.ĭalton defined the atom as the basic unit of an element that can take part in a chemical combination.The atoms that make chemical compounds are present in set proportions.That atoms of the same element are of the same type and mass.That atoms are indivisible and invisible particles.In 1803, the English scientist John Dalton, born in Cumberland, reworked Democritus' theory, as follows: In 1758, Roger Joseph Boscovich described a precursor of the atomic theory. They had no way to experiment to show whether his theory was true or not. Some other philosophers agreed, and others disagreed. Democritus thought that there was nothing between the atoms and that everything around us could be explained if we could understand how atoms worked. He thought that atoms would last forever, never change and could not be destroyed. Democritus called these small pieces of matter atoms, which means "indivisible". He said that this last piece of matter could not be cut any smaller. Democritus thought that if you cut something in half again and again, you would at last have to stop. 
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
